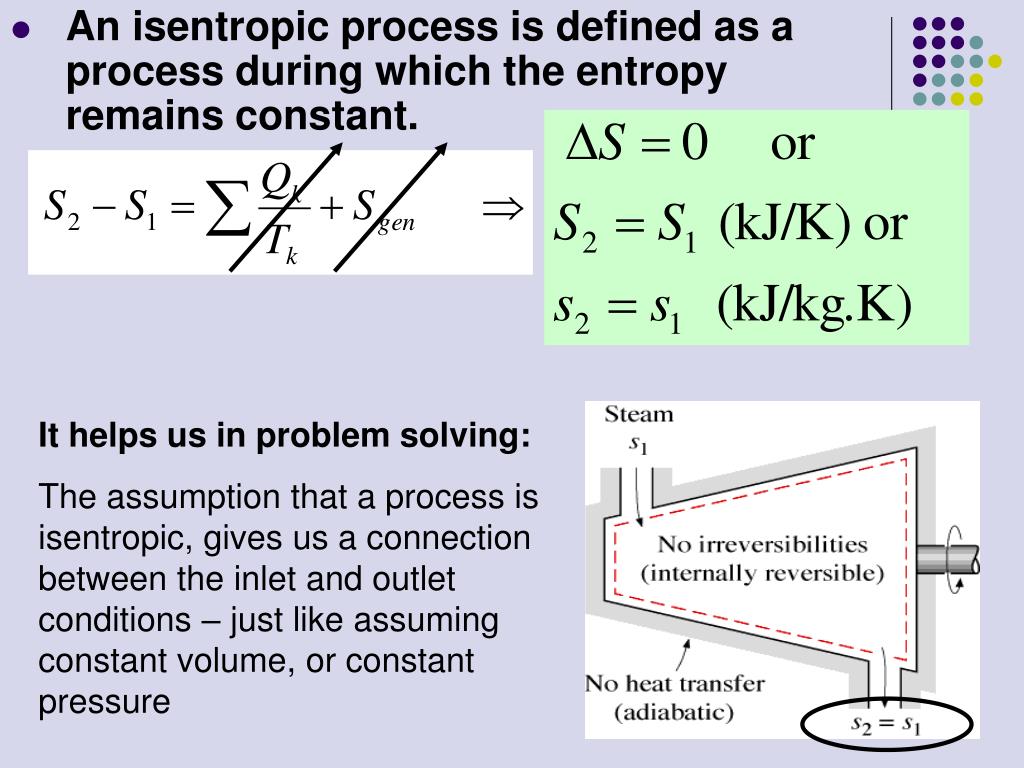
For example, the entropy change for the vaporization of water can be found as follows: So So(H 2O(g)) So(H 2O(l)) 188.7J/K mol 69.9J/K mol 118.8J/K mol. 
There might be decreases in freedom in the rest of the universe, but the sum of the increase and decrease must result in a net increase. The knowledge of the absolute entropies of substances allows us to calculate the entropy change (So) for a reaction. The freedom in that part of the universe may increase with no change in the freedom of the rest of the universe.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
